|
|||||||
| |||||||
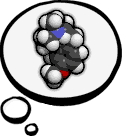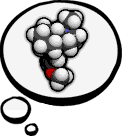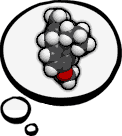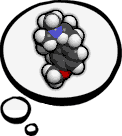
DXM-related News Dextromethorphan-related news. This particular section is publicly viewable. Feel free to post comments.
 |
|
|
Thread Tools |
|
(#1)

|
|
(#2)

|
|
||||
|
Quote:
|
 |
«
Previous Thread
|
Next Thread
»
| Thread Tools | |
|
|
Powered by vBulletin® Version 3.8.4
Copyright ©2000 - 2010, Jelsoft Enterprises Ltd.
vBulletin Skin developed by: vBStyles.com
Copyright ©2000 - 2010, Jelsoft Enterprises Ltd.
vBulletin Skin developed by: vBStyles.com
"Wiki" powered by VaultWiki v2.5.0.
Copyright © 2008 - 2010, Cracked Egg Studios.